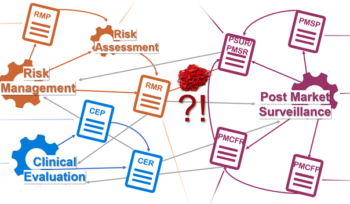
How to ensure sufficient clinical evidence for legacy devices
January 15, 2021How to claim an equivalence in the clinical evaluation under the MDR
December 21, 2020Novelties for Clinical Evaluation and Post Market Surveillance within MDR – the Posts Series
December 10, 2020Bei Medizinprodukten auf die EU warten hilft der Schweiz wenig
December 3, 2020Clinical and Performance Evaluation – our 5 strengths at your service
October 29, 2020confinis partners with b-rayZ concerning innovative Artificial Intelligence project
September 16, 2020Join us at world’s leading AI in medicine summit!
September 2, 2020Revision der Schweizer Medizinprodukteverordnung mit Folgen
August 14, 2020Workshop: Software as a Medical Device (SaMD) in Zürich
July 13, 2020Interpellation Jacqueline de Quattro on medical devices
June 2, 2020
Switzerland
- confinis ag, Hauptstrasse 16, CH-3186 Düdingen, Switzerland
- info.ch@confinis.com
- +41 26 494 8 494
- confinis corp., 4445 Willard Ave, Suite 600, Chevy Chase, MD 20815, USA
- info.usa@confinis.com
- +1 240 252 0891