How to plan your post-market clinical follow up…
- March 10, 2021
- Posted by: confinis
- Categories: confinis, Regulations
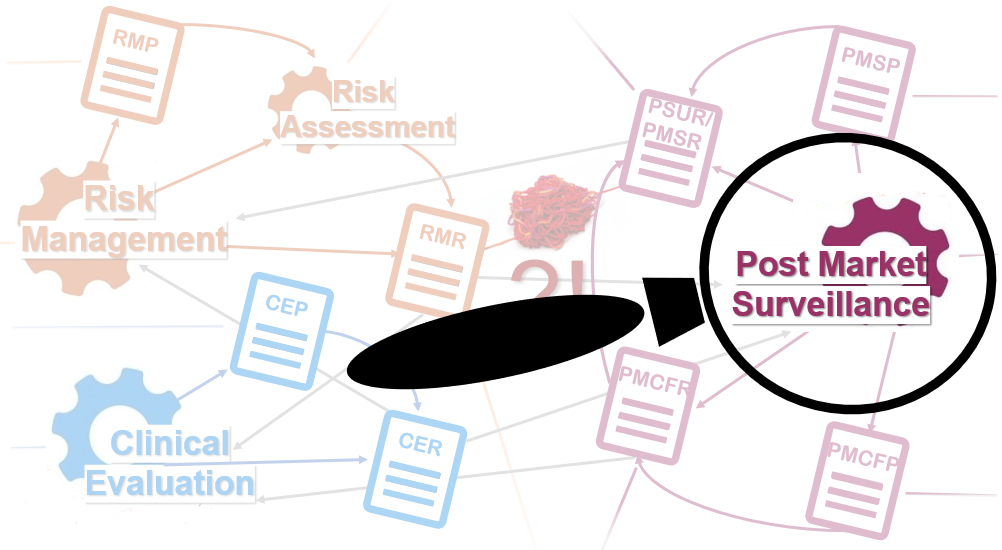
Last April, the Medical Device Coordination Group endorsed this guide for post-market clinical follow-up (PMCF) plan addressed to manufacturers and notified bodies.

It contains two main useful information:
- a contextualization of the post-market clinical follow up process with the link of the PMCF plan to post market surveillance, clinical evaluation and technical documentation;
- a template for the PMCF plan.

The PMCF plan shall be part of the Post Market Surveillance Plan, it shall set the base for the PMCF evaluation report that shall be part or an input to the Clinical Evaluation Report and the Technical Documentation.
The aims of the PMCF plan are:
- confirming the safety and performance, including the acceptability of identified risks and residual risks and the clinical benefit if applicable, of the device throughout its expected lifetime;
- identifying previously unknown side-effects and monitor the identified side-effects and contraindications;
- identifying and analysing emergent risks on the basis of factual evidence;
- ensuring the continued acceptability of the benefit-risk ratio, referred to in Section 1 and 9 of Annex I in the MDR;
- identifying possible systematic misuse or off-label use of the device, with a view to verifying that the intended purpose is correct.
The template reported in this guide for the PMCF plan proposes seven sections.
The core of the PMCF plan is the list of Activities to be conducted in post-market, described in Section C. The scope of the Activities is to obtain quality and quantity data based on the risk of the device(s) to be analysed and reported in the PMCF evaluation report. Some examples of different Activities to be conducted in the Post-Market Clinical Follow up process are: the evaluation of manufacturer device registry and national public registries, the planning of PMCF studies, real-world evidence analyses and surveys. For each Activity, the manufacturer shall provide the description of activity, define its aim, describe its different procedures, the rationale for the appropriateness of the chosen methods/procedures, and the timelines of the activity.
In conclusion, we remind you about the need to plan your Post-Market Clinical Follow Up and we provided a short overview of the MDCG 2020-7 including the contextualization of the post-market clinical follow up process and the content of the PMCF Plan.
For the full details, please refer to the MDCG 2020-7.
If you have questions or need support, please contact us at info.ch@confinis.com.
If you liked this blog and you are interested in some hints for the PMCF Evaluation Report, stay tuned for our next post in a week!
Authors: Dr. Martina Coscia, Dr. Andrea Biasiucci
Credits: the picture for this blog series has been inspired from a poster by R. Pradhan, S. Shrestha and U. Satyal entitled “Development of a digital tool for risk management, clinical evaluation and post-market surveillance of medical devices” for the 2020EuroConvergence conference.
